Hematoxylin and Eosin (H & E) staining is the most common staining technique in histopathology. This uses a combination of two dyes, Hematoxylin and Eosin used for demonstration of nucleus and cytoplasmic inclusions in clinical specimens.
Principle
Alum acts as mordant and hematoxylin containing alum stains the nucleus light blue. This turns red in presence of acid, as differentiation is achieved by treating the tissue with acid solution. Bluing step converts the initial soluble red color within the nucleus to an insoluble blue color. The counterstaining is done by using eosin which imparts pink color to the cytoplasm.
Reagents
- Harri’s Hematoxylin stain
A = 1 gm hematoxylin in 10 ml ethanol
B = 20 gm ammonium alum in hot distilled water
Mix A & B, boil and add 0.5 gm of mercuric oxide and filter.
- Eosin solution
Yellow eosin = 1 gm
Distilled water = 80 ml
Ethanol = 320 ml
Glacial Acetic Acid = 2 drops
- 0.5% HCl
- Dilute ammonia water
Procedure
- Deparaffinize the section : flame the slide on burner and place in the xylene. Repeat the treatment.
- Hydration : Hydrate the tissue section by passing through decreasing concentration of alcohol baths and water. (100%, 90%, 80%, 70%)
- Stain in hematoxylin for 3-5 minutes
- Wash in running tap water until sections “blue” for 5 minutes or less.
- Differentiate in 1% acid alcohol (1% HCl in 70% alcohol) for 5 minutes.
- Wash in running tap water until the sections are again blue by dipping in an alkaline solution (eg. ammonia water) followed by tap water wash.
- Stain in 1% Eosin Y for 10 minutes
- Wash in tap water for 1-5 minutes
- Dehydrate in increasing concentration of alcohols and clear in xylene
- Mount in mounting media
- Observe under microscope
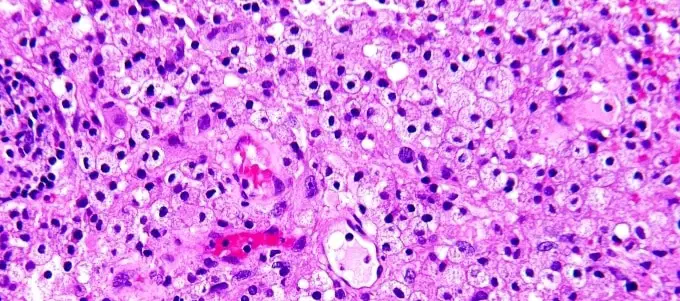
Result and Interpretation
Nuclei : blue, black
Cytoplasm : Pink
Muscle fibres : deep red
RBCs : orange red
Fibrin : deep pink

Hai i am lab technician , and also bsc(microbiology) i like your artile from many technician technician gain knowledge
Thanks
Hai this is dushyanth. This information is good for students and technicians.
thanks for your teachi ng techniques, am astudend in medical lab tech
hello, i like this we need more of these. why cant we just dewax in xylene rather than flaming, it gives good results.
Flaming before going into xylene facilitates the dewaxing process in xylene. It makes it quicker
Thid is very useful piece of info
Dear Dhurba Giri, Please contact me through my mail id [email protected]
what haematoxylin? Harris or delafield
Differentiate in 1% acid alcohol (1% HCl in 70% alcohol) for 5 minutes. from my long experience in this field I think 5 minutes it very long time only few seconds it enough to remove excess stain
One major issue…. After staining with eosin, when we put the slide in increasing grade of alcohol, the alcohol removes the eosin… What to do of that…. Kindly mail me or msg me if anyone has solution…
[email protected].
8980236234
HaI I am sivagami this information good
I want to know about staining of nucliec acid
I really like ur notes..it’s very simple nd easy to understand….
Thanks for giving good knowledge..