What is C-reactive protein (CRP)?
C-reactive protein (CRP) is a special type of protein produced by the liver in response to inflammatory cytokines such as Interleukin-6 (IL-6). CRP is classified as an acute phase reactant, which means that its levels will rise within a few hours after tissue injury, the start of an infection, or other cause of inflammation.
The most important role of CRP is its interaction with the complement system, which is one of the body’s immunologic defense mechanisms.
Why CRP test is done?
C-reactive protein (CRP) test is performed to determine if a person has a problem linked to acute infection or inflammation.
The CRP test is not diagnostic of any condition, but it can be used together with signs and symptoms and other tests to evaluate an individual for an acute or chronic inflammatory condition. These include:
- To determine if there is infection after surgery: CRP levels normally increase within two to six hours following surgery but then return to normal by the third day; if CRP levels are elevated three days after surgery it means there is an infection.
- To keep track of an infection or disease that can cause inflammation: Inflammatory bowel disease (IBD), lymphoma ( cancer of the lymph nodes), immune system diseases such as lupus (SLE), rheumatoid arthritis ( swelling of the tissues that line the joints) and osteomyelitis (infection of the bone) are some conditions in which inflammation can be monitored with a CRP test.
- To monitor treatment of a disease such as cancer or infection: Not only do CRP levels go up quickly if you have an infection but they also return to normal quickly if you are responding to the treatment.
CRP Test Principle
CRP Test is based on the latex agglutination method introduced by Singer, et. al., in 1957. This is a slide agglutination test for the qualitative and semiquantitative detection of C-Reactive Protein (CRP) in human serum.
Latex particles coated with goat IgG anti-human CRP are agglutinated when mixed with samples containing CRP. When latex particles coated with human anti-CRP are mixed with a patient’s serum containing C – reactive proteins, this results in visible agglutination wihtin 2 minutes.
CRP Test Procedure (Qualitative)
- Bring all reagents and serum sample to Room Temperature and mix latex reagent gently prior to use. Do not dilute the controls and serum.
- Place 1 drop each of serum, positive control and negative control on separate reaction circles.
- Then add CRP latex reagent 1 drop to each of the circles.
- Mix with separate mixing sticks and spread the fluid over the entire area of the cell.
- Tilt the slide back and forth slowly for 2 minutes observing preferably under artificial light.
CRP Test Procedure (Semi-Quantitative)
Sera with positive results in the screening test should be retested in the semiquantitative test for obtaining the titre.
- Make serial two fold dilutions of the sample in 9 g/L saline solution.
- Proceed for each dilution as in the qualitative method.
Interpretation
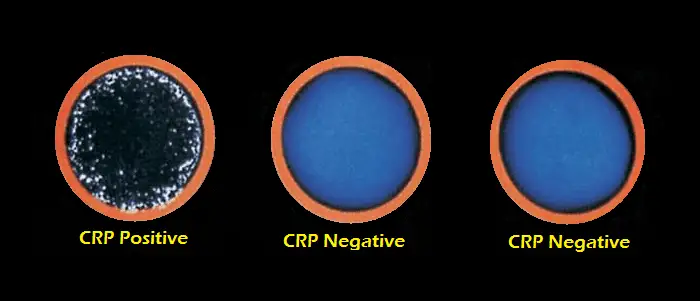
Agglutination of latex particles is considered a positive reaction, indicating the presence of C-reactive protein at a significant and detectable level. Specimens which do not contain human CRP will not cause agglutination.
If controls do not give expected reactions the test is invalid and must be repeated.
The titer, in semi-quantitative method, is defined as the highest dilution showing a positive result.

nice
great thanks!